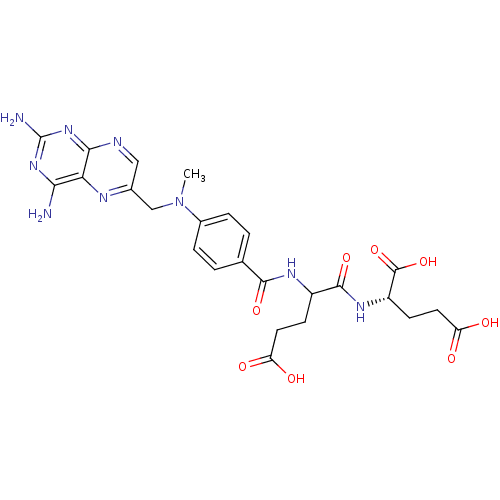
 BDBM50028534 derivative of methotrexate CHEMBL298807
BDBM50028534 derivative of methotrexate CHEMBL298807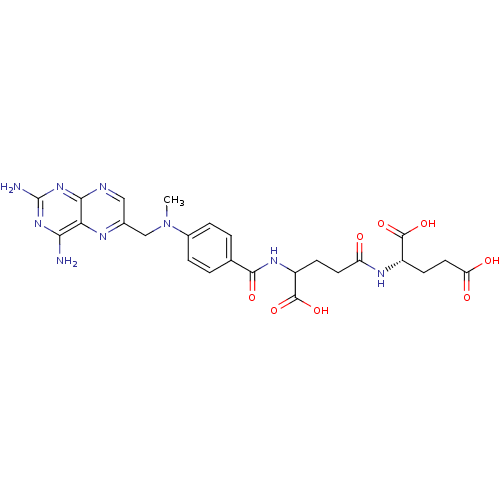 BDBM50028538 CHEMBL293147 derivative of methotrexate
BDBM50028538 CHEMBL293147 derivative of methotrexate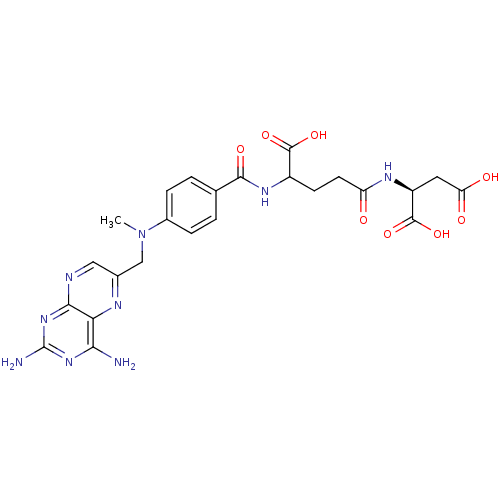 CHEMBL293546 BDBM50028542 derivative of methotrexate
CHEMBL293546 BDBM50028542 derivative of methotrexate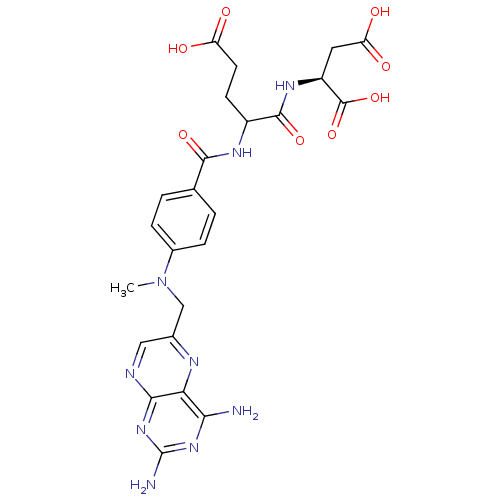 CHEMBL55797 BDBM50028535 derivative of methotrexate
CHEMBL55797 BDBM50028535 derivative of methotrexate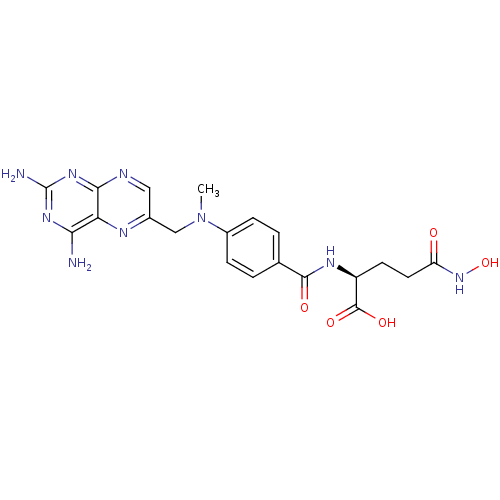 CHEMBL244883 BDBM50199731 methotrexate gamma-hydroxamic acid
CHEMBL244883 BDBM50199731 methotrexate gamma-hydroxamic acid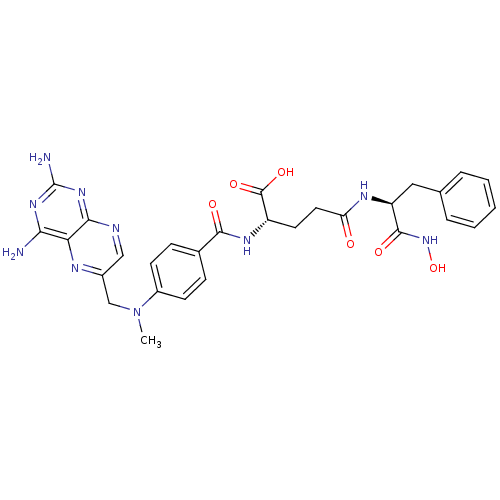 methotrexate gamma-L-phenylalaninehydroxamic acid CHEMBL396296 BDBM50199733
methotrexate gamma-L-phenylalaninehydroxamic acid CHEMBL396296 BDBM50199733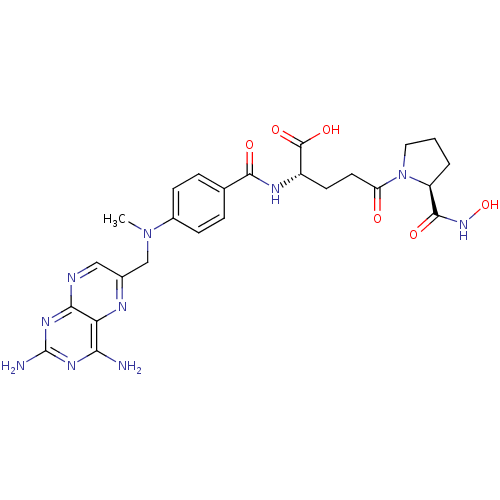 CHEMBL388879 methotrexate gamma-L-proline-hydroxamic acid BDBM50199735
CHEMBL388879 methotrexate gamma-L-proline-hydroxamic acid BDBM50199735 BDBM50600679 TCMDC-125858 TCMDC-123832 CHEMBL426 TCMDC-125488 Methotrexate
BDBM50600679 TCMDC-125858 TCMDC-123832 CHEMBL426 TCMDC-125488 Methotrexate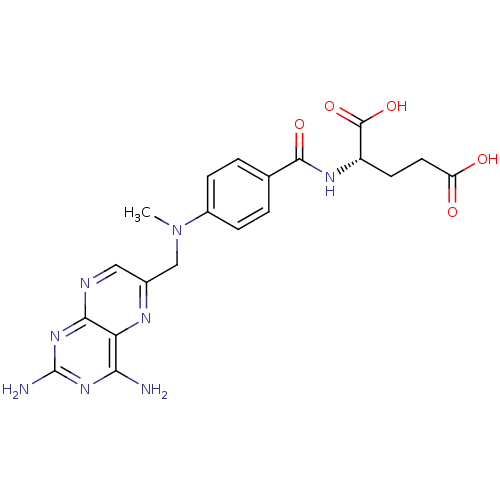 MTX BDBM18050 cid_126941 Methotrexate 2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)amino}phenyl)formamido]pentanedioic acid CHEMBL34259
MTX BDBM18050 cid_126941 Methotrexate 2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)amino}phenyl)formamido]pentanedioic acid CHEMBL34259 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Methotrexate ) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(methotrexate(MTX)) 2-(4-(((2,4-diaminopteridin-6-yl)methyl)(methyl)amino)benzamido)pentanedioic acid 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid (MTX) 2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)amino}phenyl)formamido]pentanedioic acid BDBM50004545 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Methotrexate (MTX)) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid bis-adamantan-1-ylamide 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid (Methotrexate) TREXALL 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid 4-methyl-(1S,5R,13R,14S,17R)-12-oxa-4-azapentacyclo[9.6.1.01,13.05,17.07,18]octadeca-7(18),8,10,15-tetraene-10,14-diol(methotrexate, MTX) METHOTREXATE MEXATE Methotrexate2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(MTX) FOLEX (MTX)2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid CHEMBL156 Amethopterin Abitrexate (Methotrexate, MTX)2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Methotrexate, MXT) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Amethopterin) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(methotrexate) ChEMBL_59579 (methotrexate)2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid MEXATE-AQ
2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Methotrexate ) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(methotrexate(MTX)) 2-(4-(((2,4-diaminopteridin-6-yl)methyl)(methyl)amino)benzamido)pentanedioic acid 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid (MTX) 2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)amino}phenyl)formamido]pentanedioic acid BDBM50004545 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Methotrexate (MTX)) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid bis-adamantan-1-ylamide 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid (Methotrexate) TREXALL 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid 4-methyl-(1S,5R,13R,14S,17R)-12-oxa-4-azapentacyclo[9.6.1.01,13.05,17.07,18]octadeca-7(18),8,10,15-tetraene-10,14-diol(methotrexate, MTX) METHOTREXATE MEXATE Methotrexate2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(MTX) FOLEX (MTX)2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid CHEMBL156 Amethopterin Abitrexate (Methotrexate, MTX)2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Methotrexate, MXT) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Amethopterin) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(methotrexate) ChEMBL_59579 (methotrexate)2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid MEXATE-AQ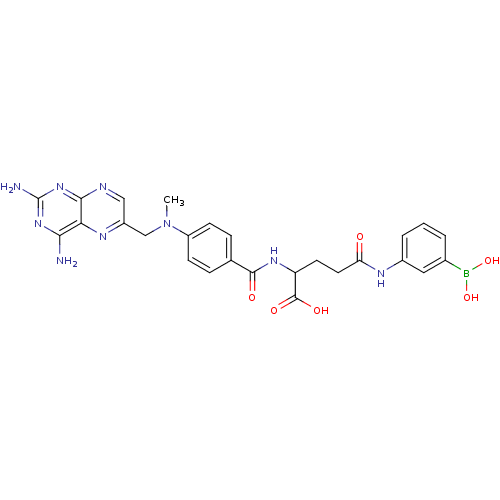 CHEMBL157117 BDBM50011321 3-(4-Carboxy-4-{4-[(2,4-diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-butyrylamino)-benzene m-boronalide(methotrexate gama-(m-boronalide))
CHEMBL157117 BDBM50011321 3-(4-Carboxy-4-{4-[(2,4-diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-butyrylamino)-benzene m-boronalide(methotrexate gama-(m-boronalide))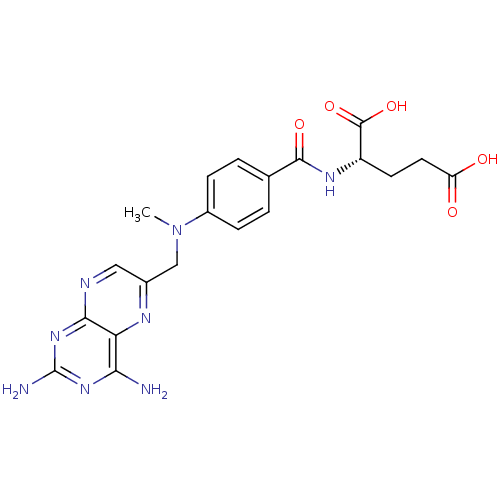 SMR000449324 cid_165528 MLS001401431 (2S)-2-[[4-[[2,4-bis(azanyl)pteridin-6-yl]methyl-methyl-amino]phenyl]carbonylamino]pentanedioic acid;hydrate (2S)-2-[[4-[(2,4-diaminopteridin-6-yl)methyl-methyl-amino]benzoyl]amino]glutaric acid;hydrate BDBM66082 L-Glutamic acid, N-[4-[[(2,4-diamino-6-pteridinyl)methyl]methylamino]benzoyl]- [CAS] (2S)-2-[[4-[(2,4-diaminopteridin-6-yl)methyl-methylamino]benzoyl]amino]pentanedioic acid;hydrate (2S)-2-[[[4-[(2,4-diamino-6-pteridinyl)methyl-methylamino]phenyl]-oxomethyl]amino]pentanedioic acid;hydrate US11530198, Example Methotrexate
SMR000449324 cid_165528 MLS001401431 (2S)-2-[[4-[[2,4-bis(azanyl)pteridin-6-yl]methyl-methyl-amino]phenyl]carbonylamino]pentanedioic acid;hydrate (2S)-2-[[4-[(2,4-diaminopteridin-6-yl)methyl-methyl-amino]benzoyl]amino]glutaric acid;hydrate BDBM66082 L-Glutamic acid, N-[4-[[(2,4-diamino-6-pteridinyl)methyl]methylamino]benzoyl]- [CAS] (2S)-2-[[4-[(2,4-diaminopteridin-6-yl)methyl-methylamino]benzoyl]amino]pentanedioic acid;hydrate (2S)-2-[[[4-[(2,4-diamino-6-pteridinyl)methyl-methylamino]phenyl]-oxomethyl]amino]pentanedioic acid;hydrate US11530198, Example Methotrexate
- Rosowsky, A; Bader, H; Radike-Smith, M; Cucchi, CA; Wick, MM; Freisheim, JH Methotrexate analogues. 28. Synthesis and biological evaluation of new gamma-monoamides of aminopterin and methotrexate. J Med Chem 29: 1703-9 (1986)
- Rosowsky, A; Bader, H; Kohler, W; Freisheim, JH; Moran, RG Methotrexate analogues. 34. Replacement of the glutamate moiety in methotrexate and aminopterin by long-chain 2-aminoalkanedioic acids. J Med Chem 31: 1338-44 (1988)
- Rosowsky, A; Forsch, RA; Yu, CS; Lazarus, H; Beardsley, GP Methotrexate analogues. 21. Divergent influence of alkyl chain length on the dihydrofolate reductase affinity and cytotoxicity of methotrexate monoesters. J Med Chem 27: 605-9 (1984)
- Pineda, P; Kanter, A; McIvor, RS; Benkovic, SJ; Rosowsky, A; Wagner, CR Dihydrofolate reductase mutant with exceptional resistance to methotrexate but not to trimetrexate. J Med Chem 46: 2816-8 (2003)
- Garland, WA; Frenzel, BD; Karg, T Methotrexate adjuvants to reduce toxicity and methods for using the same US Patent US8853227 (2014)
- Piper, JR; McCaleb, GS; Montgomery, JA; Kisliuk, RL; Gaumont, Y; Sirotnak, FM 10-Propargylaminopterin and alkyl homologues of methotrexate as inhibitors of folate metabolism. J Med Chem 25: 877-80 (1982)
- Rosowsky, A; Forsch, RA; Freisheim, JH; Galivan, J; Wick, M Methotrexate analogues. 23. Synthesis, dihydrofolate reductase affinity, cytotoxicity, and in vivo antitumor activity of some putative degradation products of methotrexate-poly(L-lysine) conjugates. J Med Chem 27: 888-93 (1984)
- Martinelli, JE; Chaykovsky, M; Kisliuk, RL; Gaumont, Y; Gittelman, MC Methotrexate analogues. 12. Synthesis and biological properties of some aza homologues. J Med Chem 22: 869-74 (1979)
- Santos, MA; Enyedy, EA; Nuti, E; Rossello, A; Krupenko, NI; Krupenko, SA Methotrexate gamma-hydroxamate derivatives as potential dual target antitumor drugs. Bioorg Med Chem 15: 1266-74 (2007)
- Masuda, M; I'izuka, Y; Yamazaki, M; Nishigaki, R; Kato, Y; Ni'inuma, K; Suzuki, H; Sugiyama, Y Methotrexate is excreted into the bile by canalicular multispecific organic anion transporter in rats. Cancer Res 57: 3506-10 (1997)
- Rosowsky, A; Forsch, RA; Wright, JE Synthesis and in vitro antifolate activity of rotationally restricted aminopterin and methotrexate analogues. J Med Chem 47: 6958-63 (2004)
- Rosowsky, A; Freisheim, JH; Moran, RG; Solan, VC; Bader, H; Wright, JE; Radike-Smith, M Methotrexate analogues. 26. Inhibition of dihydrofolate reductase and folylpolyglutamate synthetase activity and in vitro tumor cell growth by methotrexate and aminopterin analogues containing a basic amino acid side chain. J Med Chem 29: 655-60 (1986)
- Takeda, M; Khamdang, S; Narikawa, S; Kimura, H; Hosoyamada, M; Cha, SH; Sekine, T; Endou, H Characterization of methotrexate transport and its drug interactions with human organic anion transporters. J Pharmacol Exp Ther 302: 666-71 (2002)
- Uwai, Y; Saito, H; Inui, K Interaction between methotrexate and nonsteroidal anti-inflammatory drugs in organic anion transporter. Eur J Pharmacol 409: 31-6 (2001)
- Kralovec, J; Spencer, G; Blair, AH; Mammen, M; Singh, M; Ghose, T Synthesis of methotrexate-antibody conjugates by regiospecific coupling and assessment of drug and antitumor activities. J Med Chem 32: 2426-31 (1989)
- Rosowsky, A; Solan, VC; Forsch, RA; Delcamp, TJ; Baccanari, DP; Freisheim, JH Methotrexate analogues. 30. Dihydrofolate reductase inhibition and in vitro tumor cell growth inhibition by N epsilon-(haloacetyl)-L-lysine and N delta-(haloacetyl)-L-ornithine analogues and an acivicin analogue of methotrexate. J Med Chem 30: 1463-9 (1987)
- Graffner-Nordberg, M; Marelius, J; Ohlsson, S; Persson, A; Swedberg, G; Andersson, P; Andersson, SE; Aqvist, J; Hallberg, A Computational predictions of binding affinities to dihydrofolate reductase: synthesis and biological evaluation of methotrexate analogues. J Med Chem 43: 3852-61 (2000)
- Mao, Z; Pan, J; Kalman, TI Design and synthesis of histidine analogues of folic acid and methotrexate as potential folylpolyglutamate synthetase inhibitors. J Med Chem 39: 4340-4 (1996)
- DeGraw, JI; Christie, PH; Kisliuk, RL; Gaumont, Y; Sirotnak, FM Synthesis and antifolate properties of 10-alkyl-5,10-dideaza analogues of methotrexate and tetrahydrofolic acid. J Med Chem 33: 673-7 (1990)
- Chaykovsky, M; Hirst, M; Lazarus, H; Martinelli, JE Methotrexate analogues. 9. Synthesis and biological properties of some 8-alkyl-7,8-dihydro analogues. J Med Chem 20: 1323-7 (1977)
- Coats, EA; Genther, CS; Selassie, CD; Strong, CD; Hansch, C Quantitative structure-activity relationship of antifolate inhibition of bacteria cell cultures resistant and sensitive to methotrexate. J Med Chem 28: 1910-6 (1985)
- Piper, JR; Montgomery, JA; Sirotnak, FM; Chello, PL Syntheses of alpha- and gamma-substituted amides, peptides, and esters of methotrexate and their evaluation as inhibitors of folate metabolism. J Med Chem 25: 182-7 (1982)
- Rosowsky, A; Forsch, RA; Moran, RG; Freisheim, JH Synthesis and biological activity of the 2-desamino and 2-desamino-2-methyl analogues of aminopterin and methotrexate. J Med Chem 34: 227-34 (1991)
- Rosowsky, A; Bader, H; Freisheim, JH Synthesis and biological activity of methotrexate analogues with two acid groups and a hydrophobic aromatic ring in the side chain. J Med Chem 34: 574-9 (1991)
- Han, YH; Kato, Y; Haramura, M; Ohta, M; Matsuoka, H; Sugiyama, Y Physicochemical parameters responsible for the affinity of methotrexate analogs for rat canalicular multispecific organic anion transporter (cMOAT/MRP2). Pharm Res 18: 579-86 (2001)
- Rosowsky, A; Bader, H; Freisheim, JH Analogues of methotrexate and aminopterin with gamma-methylene and gamma-cyano substitution of the glutamate side chain: synthesis and in vitro biological activity. J Med Chem 34: 203-8 (1991)
- Double Methotrexate-Modified Neuropeptide Y Analogues Express Increased Toxicity and Overcome Drug Resistance in Breast Cancer Cells.
- Piper, JR; McCaleb, GS; Montgomery, JA; Kisliuk, RL; Gaumont, Y; Sirotnak, FM Syntheses and antifolate activity of 5-methyl-5-deaza analogues of aminopterin, methotrexate, folic acid, and N10-methylfolic acid. J Med Chem 29: 1080-7 (1986)
- Rosowsky, A; Forsch, RA; Bader, H; Freisheim, JH Synthesis and in vitro biological activity of new deaza analogues of folic acid, aminopterin, and methotrexate with an L-ornithine side chain. J Med Chem 34: 1447-54 (1991)
- Takeuchi, A; Masuda, S; Saito, H; Hashimoto, Y; Inui, K Trans-stimulation effects of folic acid derivatives on methotrexate transport by rat renal organic anion transporter, OAT-K1. J Pharmacol Exp Ther 293: 1034-9 (2000)
- Breedveld, P; Zelcer, N; Pluim, D; Sönmezer, O; Tibben, MM; Beijnen, JH; Schinkel, AH; van Tellingen, O; Borst, P; Schellens, JH Mechanism of the pharmacokinetic interaction between methotrexate and benzimidazoles: potential role for breast cancer resistance protein in clinical drug-drug interactions. Cancer Res 64: 5804-11 (2004)
- Rosowsky, A; Forsch, RA; Freisheim, JH; Danenberg, PV; Moran, RG; Wick, MM Methotrexate analogues. 29. Effect of gamma-aminobutyric acid spacers between the pteroyl and glutamate moieties on enzyme binding and cell growth inhibition. J Med Chem 29: 1872-6 (1986)
- Rosowsky, A; Forsch, RA; Moran, RG; Kohler, W; Freisheim, JH Methotrexate analogues. 32. Chain extension, alpha-carboxyl deletion, and gamma-carboxyl replacement by sulfonate and phosphonate: effect on enzyme binding and cell-growth inhibition. J Med Chem 31: 1326-31 (1988)
- Rosowsky, A; Bader, H; Cucchi, CA; Moran, RG; Kohler, W; Freisheim, JH Methotrexate analogues. 33. N delta-acyl-N alpha-(4-amino-4-deoxypteroyl)-L-ornithine derivatives: synthesis and in vitro antitumor activity. J Med Chem 31: 1332-7 (1988)
- Hart, BP; Haile, WH; Licato, NJ; Bolanowska, WE; McGuire, JJ; Coward, JK Synthesis and biological activity of folic acid and methotrexate analogues containing L-threo-(2S,4S)-4-fluoroglutamic acid and DL-3,3-difluoroglutamic acid. J Med Chem 39: 56-65 (1996)
- Rosowsky, A; Forsch, RA; Freisheim, JH; Moran, RG; Wick, M Methotrexate analogues. 19. Replacement of the glutamate side chain in classical antifolates by L-homocysteic acid and L-cysteic acid: effect on enzyme inhibition and antitumor activity. J Med Chem 27: 600-4 (1984)
- Selassie, CD; Li, R; Hansch, C; Khwaja, TA; Dias, CB Inhibition by 5-(substituted-benzyl)-2,4-diaminopyrimidines of murine tumor (L5178Y) cell cultures sensitive to and resistant to methotrexate. Further evidence for the sensitivity of resistant cells to hydrophobic drugs. J Med Chem 25: 518-22 (1982)
- Rosowsky, A; Bader, H; Forsch, RA; Moran, RG; Freisheim, JH Methotrexate analogues. 31. Meta and ortho isomers of aminopterin, compounds with a double bond in the side chain, and a novel analogue modified at the alpha-carbon: chemical and in vitro biological studies. J Med Chem 31: 763-8 (1988)
- Coats, EA; Genther, CS; Dietrich, SW; Guo, ZR; Hansch, C Comparison of the inhibition of methotrexate-sensitive and -resistant Lactobacillus casei cell cultures with purified Lactobacillus casei dihydrofolate reductase by 4,6-diamino-1,2-dihydro-2,2-dimethyl-1-(3-substituted-phenyl)-s-triazines. Use of quantitative structure-activity relationships in maki J Med Chem 24: 1422-9 (1981)
- ChEMBL_2215578 (CHEMBL5128710) Inhibition of human ABCG2 expressed in dog MDCK-II-BCRP cells membrane vesicles mediated transport of 3[H]-methotrexate for 5 mins using [3H]-methotrexate as substrate by radiometric scintillation analysis
- ChEMBL_2261493 (CHEMBL5216504) Inhibition of ABCG2 (unknown origin) in Sf9 membrane vesicles using [3H] methotrexate as substrate
- ChEMBL_1491712 (CHEMBL3536904) Inhibition of human BCRP-mediated methotrexate transport using inside-out membrane vesicles by LC-MS/MS analysis
- ChEMBL_1341180 (CHEMBL3256792) Competitive inhibition of thymidylate synthase purified from methotrexate-resistant Lactobacillus casei using 2'-deoxy[5-3H]uridine 5'-phosphate by radioisotope assay
- ChEMBL_2215567 (CHEMBL5128699) Inhibition of human ABCG2 expressed in human HEK293 cells membrane vesicles mediated transport of 3[H]-MTX for 2 mins using [3H]-methotrexate as substrate by rapid filtration technique
- ChEMBL_2215584 (CHEMBL5128716) Inhibition of human ABCG2 expressed in human HEK293 cells membrane vesicles mediated transport of 3[H]-MTX for 1 hr using [3H]-methotrexate as substrate by rapid filtration technique
- In Vitro Inhibition Assay Ketotifen, dacarbazine, thiocolchicoside, meloxicam, methotrexate, furosemide, olanzapine, methylprednizolone acetate, paricalcitol, ritodrine hydrochloride, and gadobenate-dimeglumine were used as inhibitors. In the media with inhibitor or without inhibitor, the substrate concentrations were 0.012 mM, 0.030 mM, 0.060 mM, 0.120 mM, and 0.450 mM. Inhibitor solutions were added to the reaction medium, resulting in three different fixed concentrations of inhibitors in 1 ml total reaction volume.
- In Vitro Assay In order to determine the effects of some drugs on human 6PGD, concentrations of ketotifen (0.0018-0.0282 mM), dacarbazine (0.0049-0.054 mM), meloxicam (0.02-0.285 mM), furosemide (0.03-0.6 mM), methotrexate (0.036-0.55 mM), metochloropramide hydrochloride (0.83-8.335 mM), ritodrine hydrochloride (1.54-15.4 mM), and gadopentetic acid (24.99-249.935 mM) were added to the reaction mixture and the enzyme activity was measured. An experiment in the absence of drug was used as control (100% activity)
 BDBM50028534 derivative of methotrexate CHEMBL298807
BDBM50028534 derivative of methotrexate CHEMBL298807 BDBM50028538 CHEMBL293147 derivative of methotrexate
BDBM50028538 CHEMBL293147 derivative of methotrexate CHEMBL293546 BDBM50028542 derivative of methotrexate
CHEMBL293546 BDBM50028542 derivative of methotrexate CHEMBL55797 BDBM50028535 derivative of methotrexate
CHEMBL55797 BDBM50028535 derivative of methotrexate CHEMBL244883 BDBM50199731 methotrexate gamma-hydroxamic acid
CHEMBL244883 BDBM50199731 methotrexate gamma-hydroxamic acid methotrexate gamma-L-phenylalaninehydroxamic acid CHEMBL396296 BDBM50199733
methotrexate gamma-L-phenylalaninehydroxamic acid CHEMBL396296 BDBM50199733 CHEMBL388879 methotrexate gamma-L-proline-hydroxamic acid BDBM50199735
CHEMBL388879 methotrexate gamma-L-proline-hydroxamic acid BDBM50199735 BDBM50600679 TCMDC-125858 TCMDC-123832 CHEMBL426 TCMDC-125488 Methotrexate
BDBM50600679 TCMDC-125858 TCMDC-123832 CHEMBL426 TCMDC-125488 Methotrexate MTX BDBM18050 cid_126941 Methotrexate 2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)amino}phenyl)formamido]pentanedioic acid CHEMBL34259
MTX BDBM18050 cid_126941 Methotrexate 2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)amino}phenyl)formamido]pentanedioic acid CHEMBL34259 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Methotrexate ) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(methotrexate(MTX)) 2-(4-(((2,4-diaminopteridin-6-yl)methyl)(methyl)amino)benzamido)pentanedioic acid 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid (MTX) 2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)amino}phenyl)formamido]pentanedioic acid BDBM50004545 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Methotrexate (MTX)) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid bis-adamantan-1-ylamide 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid (Methotrexate) TREXALL 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid 4-methyl-(1S,5R,13R,14S,17R)-12-oxa-4-azapentacyclo[9.6.1.01,13.05,17.07,18]octadeca-7(18),8,10,15-tetraene-10,14-diol(methotrexate, MTX) METHOTREXATE MEXATE Methotrexate2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(MTX) FOLEX (MTX)2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid CHEMBL156 Amethopterin Abitrexate (Methotrexate, MTX)2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Methotrexate, MXT) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Amethopterin) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(methotrexate) ChEMBL_59579 (methotrexate)2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid MEXATE-AQ
2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Methotrexate ) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(methotrexate(MTX)) 2-(4-(((2,4-diaminopteridin-6-yl)methyl)(methyl)amino)benzamido)pentanedioic acid 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid (MTX) 2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)amino}phenyl)formamido]pentanedioic acid BDBM50004545 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Methotrexate (MTX)) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid bis-adamantan-1-ylamide 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid (Methotrexate) TREXALL 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid 4-methyl-(1S,5R,13R,14S,17R)-12-oxa-4-azapentacyclo[9.6.1.01,13.05,17.07,18]octadeca-7(18),8,10,15-tetraene-10,14-diol(methotrexate, MTX) METHOTREXATE MEXATE Methotrexate2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(MTX) FOLEX (MTX)2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid CHEMBL156 Amethopterin Abitrexate (Methotrexate, MTX)2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Methotrexate, MXT) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(Amethopterin) 2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid(methotrexate) ChEMBL_59579 (methotrexate)2-{4-[(2,4-Diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-pentanedioic acid MEXATE-AQ CHEMBL157117 BDBM50011321 3-(4-Carboxy-4-{4-[(2,4-diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-butyrylamino)-benzene m-boronalide(methotrexate gama-(m-boronalide))
CHEMBL157117 BDBM50011321 3-(4-Carboxy-4-{4-[(2,4-diamino-pteridin-6-ylmethyl)-methyl-amino]-benzoylamino}-butyrylamino)-benzene m-boronalide(methotrexate gama-(m-boronalide)) SMR000449324 cid_165528 MLS001401431 (2S)-2-[[4-[[2,4-bis(azanyl)pteridin-6-yl]methyl-methyl-amino]phenyl]carbonylamino]pentanedioic acid;hydrate (2S)-2-[[4-[(2,4-diaminopteridin-6-yl)methyl-methyl-amino]benzoyl]amino]glutaric acid;hydrate BDBM66082 L-Glutamic acid, N-[4-[[(2,4-diamino-6-pteridinyl)methyl]methylamino]benzoyl]- [CAS] (2S)-2-[[4-[(2,4-diaminopteridin-6-yl)methyl-methylamino]benzoyl]amino]pentanedioic acid;hydrate (2S)-2-[[[4-[(2,4-diamino-6-pteridinyl)methyl-methylamino]phenyl]-oxomethyl]amino]pentanedioic acid;hydrate US11530198, Example Methotrexate
SMR000449324 cid_165528 MLS001401431 (2S)-2-[[4-[[2,4-bis(azanyl)pteridin-6-yl]methyl-methyl-amino]phenyl]carbonylamino]pentanedioic acid;hydrate (2S)-2-[[4-[(2,4-diaminopteridin-6-yl)methyl-methyl-amino]benzoyl]amino]glutaric acid;hydrate BDBM66082 L-Glutamic acid, N-[4-[[(2,4-diamino-6-pteridinyl)methyl]methylamino]benzoyl]- [CAS] (2S)-2-[[4-[(2,4-diaminopteridin-6-yl)methyl-methylamino]benzoyl]amino]pentanedioic acid;hydrate (2S)-2-[[[4-[(2,4-diamino-6-pteridinyl)methyl-methylamino]phenyl]-oxomethyl]amino]pentanedioic acid;hydrate US11530198, Example Methotrexate